Companies: Cochlear Recalls, Nufarm Settles
Now isn't a good time to be announcing bad news, or news that the jittery investors take as being bad.
We saw that last month with surfwear maker Billabong which reported lower earnings and withdrew previous guidance: the shares fell 26%.
As well, shares in BlueScope Steel fell by a quarter over several days after revealing plans to write $1 billion of asset values, then confirming those plans and revealing job cuts of 1,000, plus two plant closures.
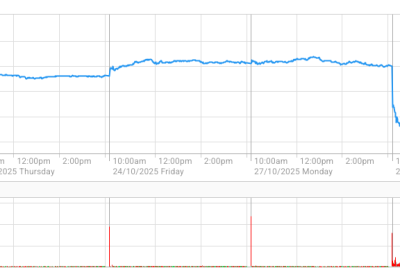
Yesterday hearing implant company Cochlear saw its shares down 27% at one stage after it announced a voluntary recall of its unimplanted CI500 Cochlear range.
The shares dropped $19.41, or 27 % to $52.77.and closed down 21% or $14.86, at $57.32, their lowest for more than two years.
The Sydney-based company said in the announcement to the ASX that it had begun notifying healthcare professionals and regulatory authorities about the recall.
Cochlear said the financial impact of the recall was difficult to forecast at this stage and CEO Chris Roberts told a conference call that "We don't know what the cause is".
"That's exactly what we need to evaluate.
"We want to avoid saying it's a or b or c when we have to up every stone and examine every possibility," he said.
In the statement, the company said: "While less than 1% of CI512 implants have failed since launch in 2009, Cochlear has identified a recent increase in the number of Nucleus CI512 implant failures.
"In an abundance of caution and with our recipients in mind, Cochlear has issued a voluntary recall of the Nucleus CI500 range of cochlear implants while it further investigates the issue.
"No implants outside the Cochlear Nucleus CI500 range are affected by this voluntary recall including Nucleus Freedom implants which remain available.
"The Nucleus 5 external devices (Nucleus CP810 Sound Processor) and any previous generation sound processors are not affected by this recall."
"Cochlear said less than 1 per cent of CI512 implants had failed since its launch in 2009, but that it had identified a recent increase in the number of Nucleus CI512 implant failures.
"Cochlear has implanted its bionic ear device in more than 250,000 people over the past 30 years.
"The company said the affected CI500 implant range includes the Nucleus CI512 implant plus the following implants which are only available in limited markets - Nucleus CI513, Nucleus CI551 double array implant and Nucleus ABI 541 Auditory Brainstem Implant.
"Its Nucleus 5 external device range and any other previous generation externals are not subject to this recall.
"If a Nucleus CI512 failure occurs, the implant safely shuts down. In the event of a failure, recipients can be re-implanted with the Nucleus Freedom implant range which remains available. Nucleus Freedom is fully compatible with the Nucleus 5 Sound Processor System which is not affected by this recall.
"For people considering a cochlear implant, Cochlear recommends the use of the Nucleus Freedom range of Cochlear implants which are fully compatible with the Nucleus 5 sound processor system. This combination of Nucleus Freedom and Nucleus 5 sound processor system does not sacrifice any of the performance benefits of the Nucleus 5 processor system.
"For more than 25 years, cochlear implant reliability has been of paramount importance to Cochlear, and we remain committed to providing high quality, high reliable cochlear implant systems.
"We sincerely apologise for any concern this causes," the company ended its statement.
The herbicide and pesticide manufacturer and importer, Nufarm said yesterday that its underlying 2011 operating profit will exceed the top end of its $88 million to $94 million guidance range.
That will be after the costs associated with the settlement of a long running dispute over receivables. Managing director Doug Rathbone said the settlement proposal is, on balance, acceptable to its board and has allowed the matter to be finalised. "The company gave detailed consideration to a number of matters in arriving at the decision to settle the dispute, including the management time that would have been consumed by further hearings to fully and finally determine the issues between the parties; litigation risk; and the ongoing commercial relationship between the parties," he said in the statement.
The company said the total write-down associated with the unrecovered portion of the receivable will be treated as a material item in Nufarm's 2011 full year accounts and will reduce net profit by $28 million. The company said its full-year results for the 12 months to July 31, 2011 will be released on September 28 and are still subject to final audit.
The company reported an operating profit of $58.6 million for fiscal 2010 and a net loss of $22.6 million after a series of write-downs.
The shares fell 6c to $3.89, not a bad outcome in yesterday's bloodbath.
Copyright Australasian Investment Review.
AIR publishes a weekly magazine. Subscriptions are free at www.aireview.com.au
Now isn't a good time to be announcing bad news, or news that the jittery investors take as being bad.
We saw that last month with surfwear maker Billabong which reported lower earnings and withdrew previous guidance: the shares fell 26%.
As well, shares in BlueScope Steel fell by a quarter over several days after revealing plans to write $1 billion of asset values, then confirming those plans and revealing job cuts of 1,000, plus two plant closures.
Yesterday hearing implant company Cochlear saw its shares down 27% at one stage after it announced a voluntary recall of its unimplanted CI500 Cochlear range.
The shares dropped $19.41, or 27 % to $52.77.and closed down 21% or $14.86, at $57.32, their lowest for more than two years.
The Sydney-based company said in the announcement to the ASX that it had begun notifying healthcare professionals and regulatory authorities about the recall.
Cochlear said the financial impact of the recall was difficult to forecast at this stage and CEO Chris Roberts told a conference call that "We don't know what the cause is".
"That's exactly what we need to evaluate.
"We want to avoid saying it's a or b or c when we have to up every stone and examine every possibility," he said.
In the statement, the company said: "While less than 1% of CI512 implants have failed since launch in 2009, Cochlear has identified a recent increase in the number of Nucleus CI512 implant failures.
"In an abundance of caution and with our recipients in mind, Cochlear has issued a voluntary recall of the Nucleus CI500 range of cochlear implants while it further investigates the issue.
"No implants outside the Cochlear Nucleus CI500 range are affected by this voluntary recall including Nucleus Freedom implants which remain available.
"The Nucleus 5 external devices (Nucleus CP810 Sound Processor) and any previous generation sound processors are not affected by this recall."
"Cochlear said less than 1 per cent of CI512 implants had failed since its launch in 2009, but that it had identified a recent increase in the number of Nucleus CI512 implant failures.
"Cochlear has implanted its bionic ear device in more than 250,000 people over the past 30 years.
"The company said the affected CI500 implant range includes the Nucleus CI512 implant plus the following implants which are only available in limited markets - Nucleus CI513, Nucleus CI551 double array implant and Nucleus ABI 541 Auditory Brainstem Implant.
"Its Nucleus 5 external device range and any other previous generation externals are not subject to this recall.
"If a Nucleus CI512 failure occurs, the implant safely shuts down. In the event of a failure, recipients can be re-implanted with the Nucleus Freedom implant range which remains available. Nucleus Freedom is fully compatible with the Nucleus 5 Sound Processor System which is not affected by this recall.
"For people considering a cochlear implant, Cochlear recommends the use of the Nucleus Freedom range of Cochlear implants which are fully compatible with the Nucleus 5 sound processor system. This combination of Nucleus Freedom and Nucleus 5 sound processor system does not sacrifice any of the performance benefits of the Nucleus 5 processor system.
"For more than 25 years, cochlear implant reliability has been of paramount importance to Cochlear, and we remain committed to providing high quality, high reliable cochlear implant systems.
"We sincerely apologise for any concern this causes," the company ended its statement.
The herbicide and pesticide manufacturer and importer, Nufarm said yesterday that its underlying 2011 operating profit will exceed the top end of its $88 million to $94 million guidance range.
That will be after the costs associated with the settlement of a long running dispute over receivables. Managing director Doug Rathbone said the settlement proposal is, on balance, acceptable to its board and has allowed the matter to be finalised. "The company gave detailed consideration to a number of matters in arriving at the decision to settle the dispute, including the management time that would have been consumed by further hearings to fully and finally determine the issues between the parties; litigation risk; and the ongoing commercial relationship between the parties," he said in the statement.
The company said the total write-down associated with the unrecovered portion of the receivable will be treated as a material item in Nufarm's 2011 full year accounts and will reduce net profit by $28 million. The company said its full-year results for the 12 months to July 31, 2011 will be released on September 28 and are still subject to final audit.
The company reported an operating profit of $58.6 million for fiscal 2010 and a net loss of $22.6 million after a series of write-downs.
The shares fell 6c to $3.89, not a bad outcome in yesterday's bloodbath.
Copyright Australasian Investment Review.
AIR publishes a weekly magazine. Subscriptions are free at www.aireview.com.au